An acyl anion is unstable on its own, but can be very stable if we use TMSCN to temporarily mask it.
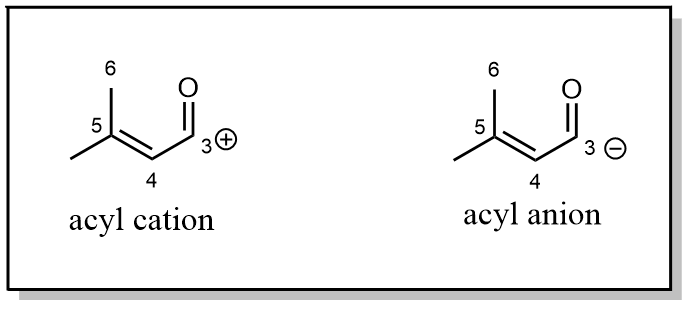
The carbonyl group is a strong electrophile; it is often recognised as a partially positively charged group. But from a retrosynthetic plan, people found they already had an electrophile, so the acyl group had to act like an anion that needs to be partially negatively charged.
Now let’s have a look at a simple retrosynthesis of a compound. There are many potential reactions we can apply here to obtain this compound. The simplest one may be the aldol condensation, if we break it down between carbon 4 and 5.
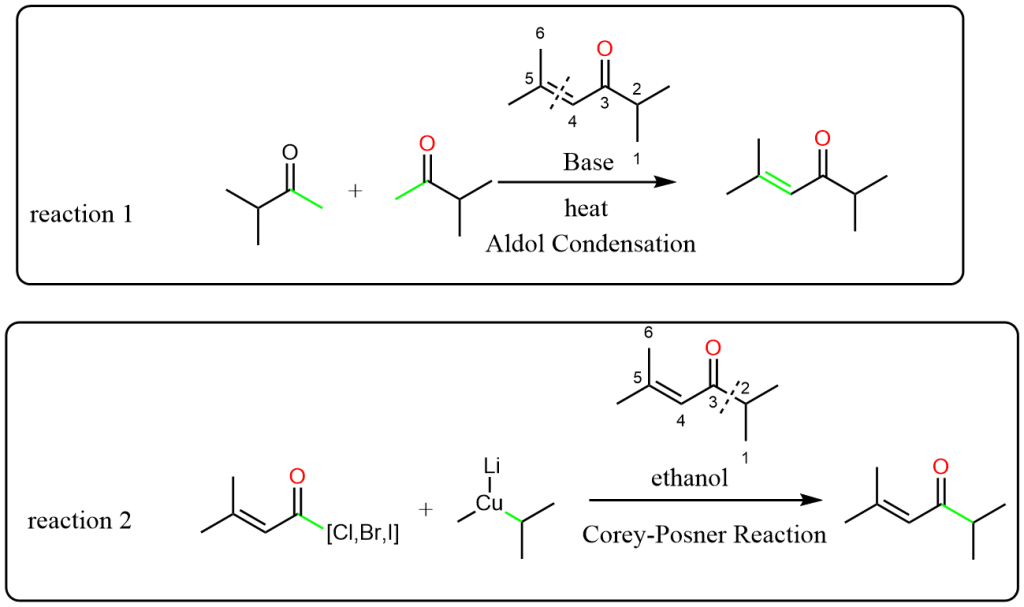
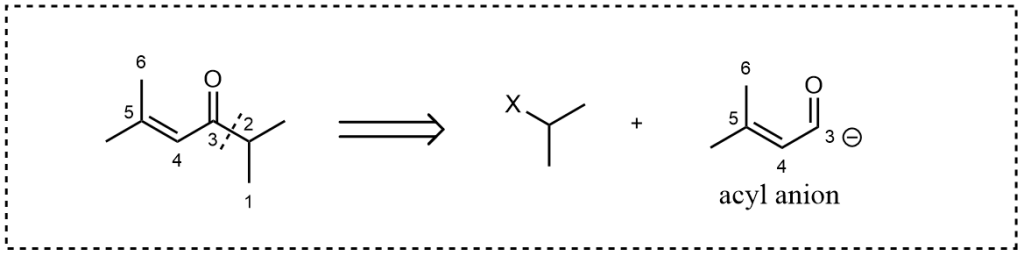
If we decide to break down carbon 3 and 2, things begin to get a bit complex. If you want to use metal, a reaction such as the Corey-Posner Reaction might be applicable. But what if you don’t want to use metal?
We need a potential reaction 3 as shown below. One can assume that the right-hand side subunit is an alkyl halogen, as an electrophile, while the left-hand side is an acyl anion. The question is how we could obtain such a species in practice, given the acyl group’s positive charge.
One way to do this is to take TMSCN as a masking group by addition to the acyl group, which can then be deprotonated, serving as a negatively charged nucleophile instead of the electrophile. After nucleophilic addition to an alkyl halide, it is unmasked back into an acyl group.
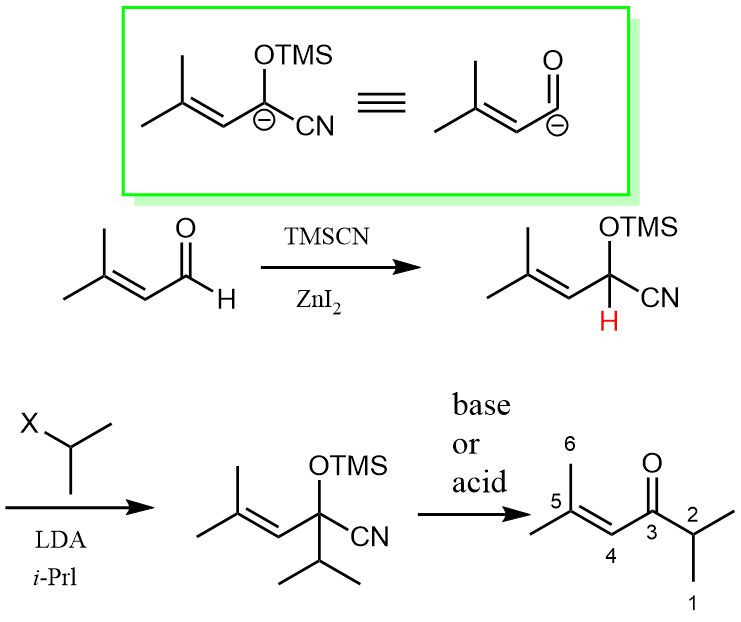
As the green box above shows, we treat the TMSCN-masked anion as an acyl anion equivalent in practice. The reason why this could work nicely is partly due to the strong electron-withdrawing nature of the CN group, which can stabilise the negative charge after LDA deprotonation.
This strategy was applied in the total synthesis of (-)-Curvulamine by the Maimone Group at UC, Berkeley. We only show part of their synthesis plan that involves our topic here.
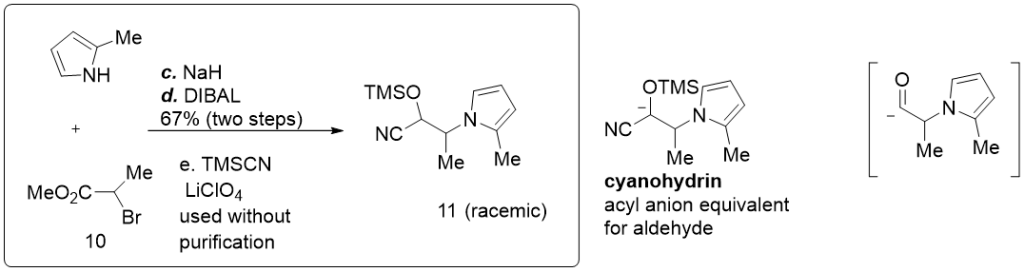
What they need is an acyl group similar to the one we discussed above. They also adopted the same strategy, using TMSCN as a masking group, in a three-step one-pot reaction:
- First, an SN2 alkylation to the 2-methyl pyrrole N atom,
- Then a reduction from ester to aldehyde,
- Followed by the TMSCN masking.
For the whole process of Curvulamine synthesis, you can read the total synthesis of (-)-Curvulamine.